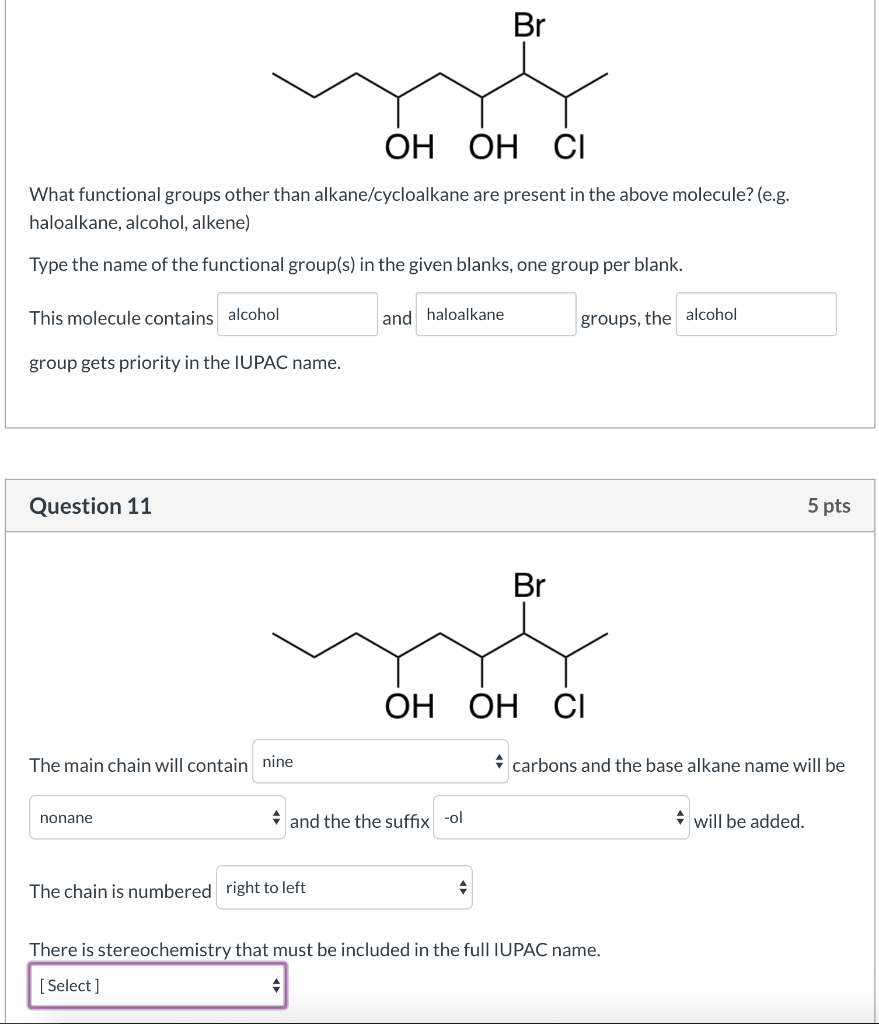
Here, we report a new radical-mediated intermolecular 1,2-dialkylation of terminal alkenes with two distinct C(sp 3)─H bonds enabled by synergistic photoredox catalysis and iron catalysis ( Fig. We envisioned that using photoredox and transition metal catalysts cooperatively might allow the controlled and simultaneous transformation of an alkene with two or more C(sp 3)─H bonds under mild conditions by regulating the oxidation and reduction potentials. In these radical processes, selectivity and reactivity could be manipulated mainly through tuning the oxidation and reduction potentials of the reaction partners and catalysts ( 29– 32). Merging visible light photoredox catalysis with transition metal catalysis has become a conceptually powerful strategy for the formation of chemical bonds owing to its extraordinary catalytic activity and mild conditions ( 29– 32), and this method has also been applied to the functionalization of C(sp 3)─H bonds ( 29– 37). Moreover, examples that allow the addition of alkyl groups across alkenes to achieve dicarbofunctionalization are rare this is partly because competitive side reactions, such as the Heck-type β-hydride elimination, homocoupling, isomerization, and/or protodemetalation, are common in these types of reactions ( 5– 18). However, the vast majority of alkene dicarbofunctionalization approaches focus on the merger of the classical cross-couplings ( 1– 18), which suffer from requiring expensive nucleophilic and electrophilic functional reagents, such as organometallic species and organohalides, for inserting the C═C bonds in the presence of noble transition metals and/or ligands. Typical technologies include alkene dicarbofunctionalizations that enable the concomitant incorporation of two vicinal carbon-based functional groups to lengthen carbon chains and increase structural complexity ( 1– 4). In this field, the functionalization of a molecule through transformations of an alkene and/or C─H bonds is a particularly fascinating but challenging goal, which has therefore attracted substantial attention from chemical researchers. Therefore, the proposed name cannot be used.The development of a mild, economical, and practical catalytic process that can selectively and rapidly increase molecular complexity starting from readily available materials, especially petroleum-based feedstocks (e.g., alkenes, alkanes, and arenes), is a critical objective in both the academic and industrial communities. The correct name for this compound is pent-2-en-2-ol (the following structure diagram shows the 2 Z isomer).Īnyway, the proposed name 2-hydroxypent-2-ene as well as the conforming name pent-2-en-2-ol do not describe the structure that is shown in the question (note the position of the double bond). Hence, this nonconforming name could still be understood. Nevertheless, this name is unambiguous it describes only one compound (ignoring cis/ trans isomerism).


The alternative name 2-hydroxypent-2-ene that is proposed in the question is not in accordance with current or past IUPAC recommendations therefore, it should not be used. Hence, the proposed name could be understood. However, the very similar name 3-penten-2-ol (note the elision of the ‘e’) was in accordance with the obsolete 1979 IUPAC recommendations. The first name 3-pentene-2-ol that is proposed in the question is not in accordance with current IUPAC recommendations. deviations from the rules for the priority of suffixes, numbering of locants, or order of citation in the name) do not necessarily lead to wrong structures.
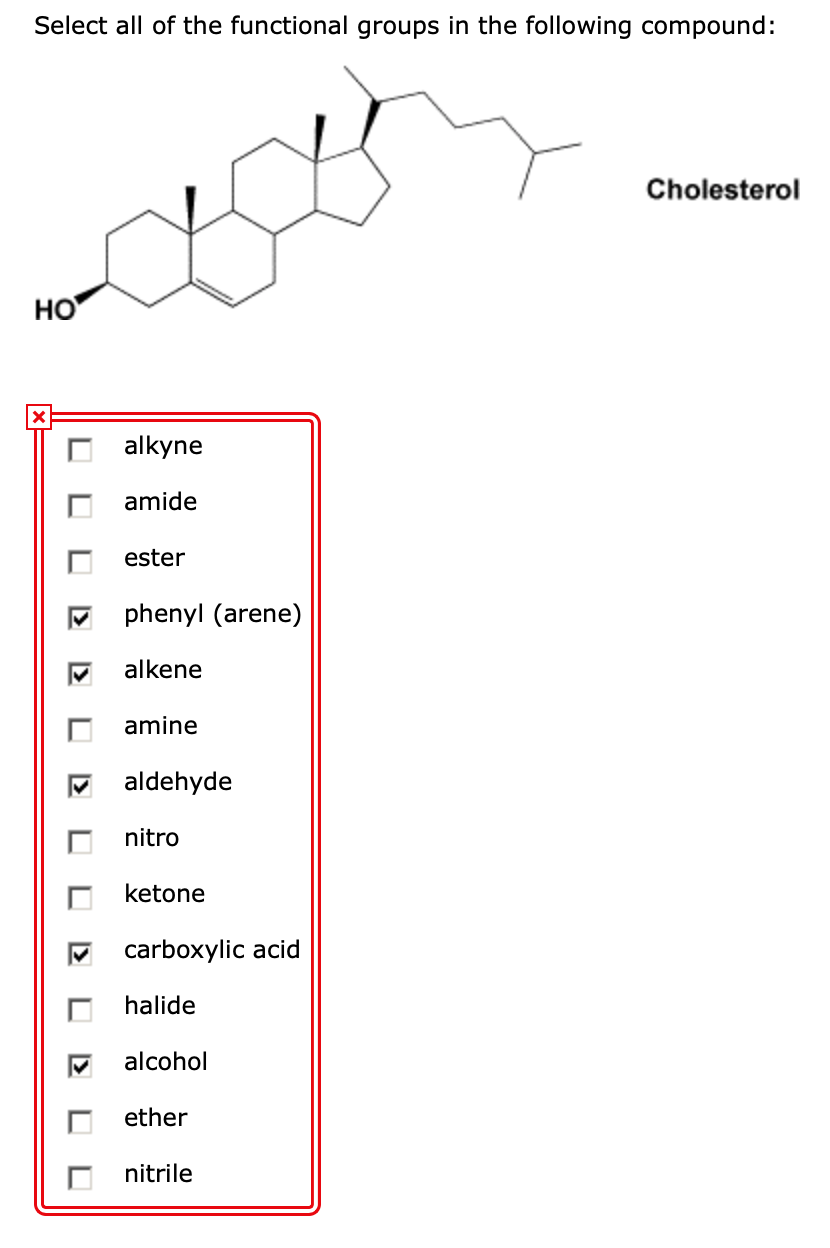
Other names that are in accordance current IUPAC recommendations and also many names that were generated according to earlier recommendations may be used in general nomenclature.įurthermore, the IUPAC nomenclature is quite robust. The PIN for the compound that is given in the question is (3 E)-pent-3-en-2-ol. Therefore, the current version of Nomenclature of Organic Chemistry – IUPAC Recommendations and Preferred Names 2013 (Blue Book) include the definition of a preferred IUPAC name (PIN), which is the name preferred among two or more names generated from two or more IUPAC recommendations including the many retained names. Generally, since many compounds can have two or more names in accordance with several methods recommended by IUPAC, a compound may be named correctly in more ways than one.
